Why Spreadsheets Fail At Scale In Quality & Compliance Management
Managing quality and compliance through spreadsheets often feels practical in the early stages. Spreadsheets are familiar, easy to deploy, and flexible enough to track documents, audits, risks, corrective actions, and training records at a basic level.
As organisations grow, however, quality and compliance requirements expand across processes, teams, locations, and regulatory frameworks. Internal audits, certification audits, customer audits, and regulatory assessments increasingly depend on data integrity, controlled workflows, traceability, and continuous audit readiness.
n many organisations, quality and compliance management continues to rely on spreadsheets instead of structured digital document management systems (DMS), quality management software, or compliance tracking software. While spreadsheets may appear sufficient at an early stage, they introduce structural limitations as operational scope increases. These limitations rarely appear immediately. They develop gradually and become visible only when audit preparation becomes time-consuming, data reliability is questioned, or compliance activities begin to rely heavily on manual coordination rather than controlled processes.
As organisational complexity increases, spreadsheet-based methods struggle to support consistent control, traceability, and audit readiness across quality and compliance functions.
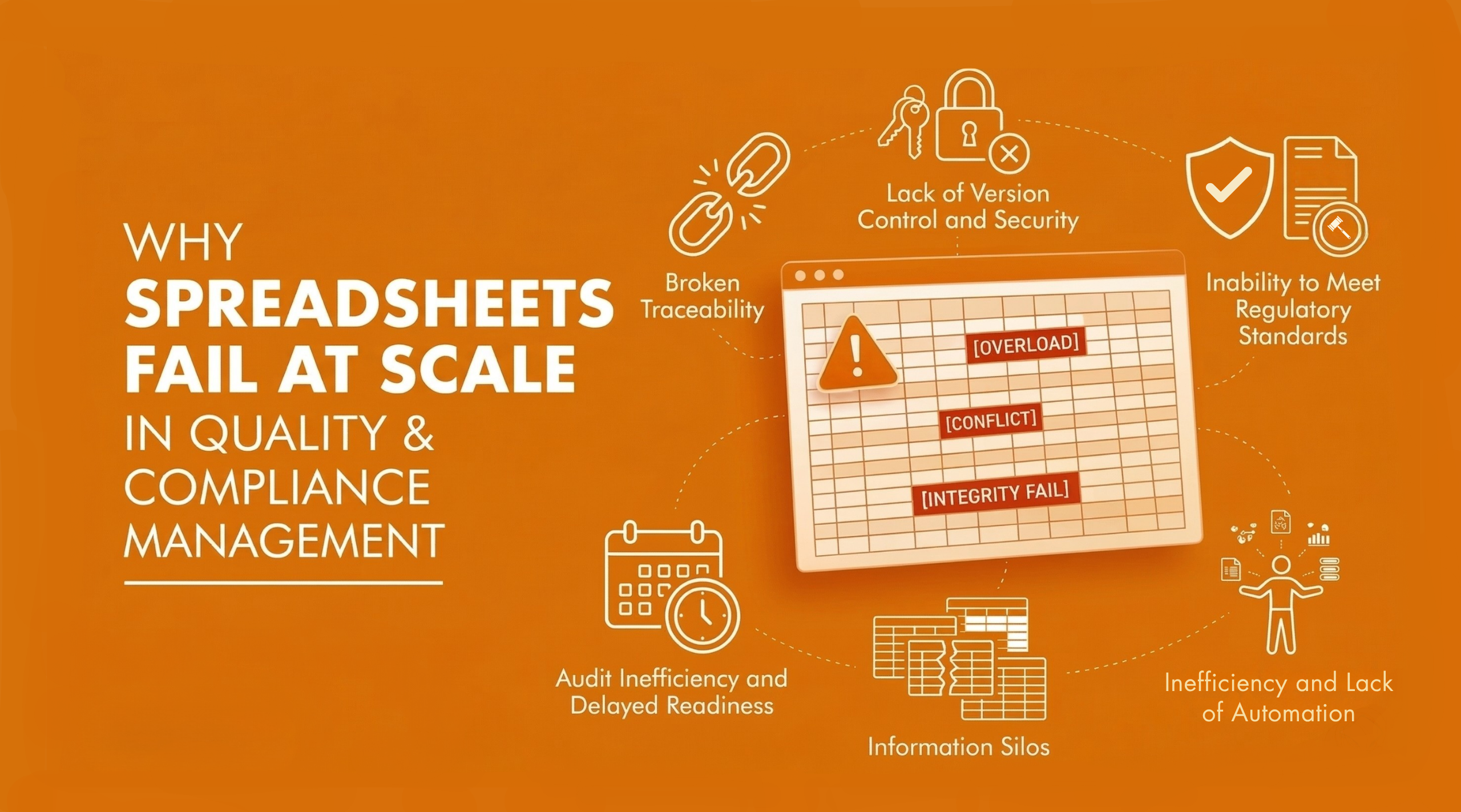
Key Signs Your Quality & Compliance Processes Have Outgrown Spreadsheets
As quality systems mature, spreadsheet-based compliance management begins to show structural limitations. The following signs indicate when spreadsheets are no longer capable of supporting scale, consistency and audit-ready governance. When multiple indicators appear together, they signal the need to move beyond spreadsheets toward structured, system-driven control.
-
Inherent Data Integrity and Error Risks:
Spreadsheets rely heavily on manual data entry, formulas, and user discipline. As data volumes grow, the likelihood of errors increases. Typographical mistakes, broken formulas, overwritten cells, and copy-paste errors often go unnoticed but directly impact the reliability of compliance records.
Unlike quality management software or audit-ready software, spreadsheets provide limited data validation. Incorrect data types, missing fields, and inconsistent formats are accepted without restriction. Over time, this weakens confidence in risk registers, audit findings, CAPA tracking, and management review inputs.
In asset and compliance registers, manual spreadsheet maintenance frequently leads to outdated or inactive records remaining active. Retired or obsolete items continue to appear valid, undermining traceability. When foundational data integrity is compromised, every downstream quality control becomes less effective.
-
Lack of Version Control and Security:
As more users interact with spreadsheets, version control becomes increasingly difficult to manage. Files are duplicated, renamed, emailed and stored across shared folders, creating multiple parallel versions with no clear source of truth.
In a spreadsheets vs DMS comparison, this is a critical limitation. Spreadsheets do not enforce document lifecycle stages such as drafting, review, approval, and controlled release. Obsolete versions often remain accessible, and approval evidence is fragmented or missing.
Spreadsheets also lack granular, role-based access control. Unlike enterprise-grade digital document management or quality management software, sensitive compliance data can be edited, deleted, or shared unintentionally. This increases the risk of unauthorised changes, data loss, and audit exposure.
-
Inability to Meet Regulatory Standards:
Many regulatory and certification standards expect capabilities that spreadsheets are not designed to provide. Frameworks such as ISO 9001, ISO 13485, GMP, and FDA 21 CFR Part 11 require controlled audit trails, documented change history, and verifiable approvals.
Spreadsheet-based systems do not provide reliable audit trails or enforce electronic approvals. Change tracking is inconsistent and often overwritten. Manual logs or comments do not meet regulatory expectations for traceability.
Unlike compliance tracking software or audit-ready software, spreadsheets force organisations to explain compliance through process descriptions rather than demonstrate it through system behaviour. During audits, this leads to deeper scrutiny, extended sampling, and increased non-conformity risk.
-
Limited Scalability and Performance
Spreadsheets are not designed to scale with growing compliance demands. As datasets expand, files become slow, unstable, and prone to crashing. Structural limits are reached quickly in environments managing documents, audits, suppliers, CAPAs, and training records together.
Complex formulas linking multiple sheets or files introduce fragile dependencies. Minor structural changes often break calculations without warning. These failures frequently surface during audits or reporting deadlines, when recovery time is limited.
In contrast, quality management software and structured digital document management systems are designed to scale without degrading performance. A compliance system that becomes unreliable as scale increases introduces operational and audit risk.
-
Inefficiency and Lack of Automation
Quality and compliance processes require timely execution and follow-up. In spreadsheet-driven environments, actions depend on emails, reminders, and individual follow-through rather than automated workflows.
Spreadsheets cannot trigger approvals, notifications, escalations, or status changes based on defined rules. Unlike audit-ready software, they do not enforce accountability or deadlines. Corrective actions are delayed, effectiveness checks are missed, and recurring issues remain unresolved.
As audit scope and regulatory expectations increase, manual coordination adds workload without improving compliance control. Effort increases, but governance does not.
-
Information Silos
Spreadsheets encourage fragmented ownership of compliance data. Different departments and locations maintain separate files using varying formats, terminology, and assumptions. Quality, EHS, operations, procurement, and maintenance data remain siloed.
Without integrated digital document management or compliance tracking software, consolidating information for audits or management reviews becomes time-consuming and error-prone. Leadership receives delayed and partial visibility into compliance status, limiting the ability to identify cross-functional trends or systemic risks. Governance becomes reactive rather than proactive. Issues are addressed locally instead of at an organisational level.
Spreadsheets do not fail suddenly. They do not alert, restrict or enforce controls. Problems accumulate quietly and surface only during audits, customer assessments, or regulatory inspections.
At that point, organisations respond reactively reconciling data, recreating evidence and validating records manually. This increases effort and stress without addressing the underlying structural limitation.
What Is The Right Solution When Spreadsheets No Longer Scale?
When spreadsheet-based methods begin to limit data integrity, traceability, and audit readiness, the solution is not to improve spreadsheet discipline or add more manual controls. These measures increase effort but do not resolve the structural limitations inherent in spreadsheet-driven systems.
The appropriate solution is to transition core quality and compliance activities to structured, system-driven platforms that are designed specifically to support governance, regulatory requirements, and audit expectations at scale. Such systems replace manual coordination with enforced process control and continuous evidence capture.
An effective solution must address all core compliance needs together, not in isolation.
-
Digital document management for controlled document lifecycle, versioning, approvals, and access control
A structured digital document management system ensures that documents are created, reviewed, approved, released, revised, and retired within a controlled lifecycle. Version control is enforced by the system rather than managed through file naming or manual discipline. Only approved and current documents remain accessible for operational use.
Access is governed by defined roles and responsibilities, preventing unauthorised edits or accidental deletions. Obsolete documents are automatically withdrawn from use, and approval history remains permanently traceable. This level of control is essential for maintaining document integrity and demonstrating compliance during audits.
-
Quality management functionality for audits, non-conformities, corrective actions, and effectiveness verification
Quality management systems must support the full audit lifecycle, from planning and execution to findings, corrective actions, and closure. Audit findings should be captured in a structured manner and directly linked to corrective and preventive actions.
Ownership, timelines, and verification requirements must be enforced consistently across all actions. Effectiveness checks should be mandatory before closure to prevent recurrence of issues. Managing these activities within a single system ensures that audits drive improvement rather than generating disconnected records.
-
Compliance tracking to maintain traceability across risks, controls, actions and evidence
Compliance tracking requires clear, continuous traceability between identified risks, implemented controls, audit findings, corrective actions, and supporting evidence. This traceability must be maintained automatically as part of normal operations, not recreated during audits.
A structured system preserves these relationships across records, allowing organisations to demonstrate compliance logically and consistently. During audits or regulatory reviews, evidence can be traced back to its source without manual reconciliation, reducing uncertainty and audit exposure.
-
Audit-ready system behaviour that preserves continuous evidence
Audit readiness should not depend on last-minute data collection or manual validation. An audit-ready system captures approvals, revisions, actions, and closures automatically as activities occur.
This creates a continuous audit trail that reflects actual system behaviour over time. When audits take place, evidence already exists in a complete, traceable, and verifiable form. This reduces preparation effort, avoids reactive fixes, and supports consistent audit outcomes across standards and locations.
How Pyraman Supports Audit-Ready Quality & Compliance Management
Pyraman is built as an all-in-one platform to manage ISO and management system requirements in a structured, audit-ready manner. Instead of using spreadsheets or disconnected tools, it brings documents, audits, and corrective actions into one controlled system to ensure consistency and traceability.
Within this framework, Pyraman enables organisations to:
- Centralise ISO documentation under controlled workflows.
- Maintain version control with defined approvals.
- Link audits directly to corrective actions.
- Track non-conformances with clear ownership.
- Preserve evidence automatically for audit traceability.
The platform integrates document management, audit tracking, corrective actions, training records and compliance documentation into one connected environment. This supports continuous audit readiness rather than last-minute preparation.
Operationally, this results in:
- Reduced manual coordination.
- Real-time visibility of compliance status.
- Faster audit preparation.
- Improved accountability across teams.
- Structured, repeatable compliance processes.
By centralising quality and compliance activities, Pyraman helps organisations move from reactive compliance to controlled, system-driven management. Ready to strengthen your audit preparedness? Contact us to see how Pyraman can support your quality and compliance requirements.
Frequently Asked Questions
-
Why do spreadsheets fail in quality and compliance management as organisations scale?
Spreadsheets rely on manual updates, individual discipline, and informal controls. As data volume, audit frequency, and regulatory scope increase, this approach leads to errors, version conflicts, and weak traceability. These limitations reduce audit readiness and increase the effort required to demonstrate compliance.
-
How does spreadsheets vs DMS impact audit outcomes?
Digital Document Management Systems enforce version control, approvals, and access restrictions by system design. Spreadsheets depend on naming conventions and manual checks, which auditors often challenge. A DMS provides clearer evidence of document control during audits.
-
When should an organisation move to quality management software?
When audits, non-conformities, corrective actions, and training records can no longer be managed consistently through spreadsheets. Increasing audit findings, delayed closures, or repeated manual reconciliation are common indicators that structured quality management software is needed.
-
Is compliance tracking software mandatory for ISO certification?
Compliance tracking software is not mandatory for ISO certification. However, it significantly improves traceability, consistency, and audit preparedness, especially in multi-process or regulated environments where manual tracking becomes unreliable.
-
What makes a system audit-ready?
An audit-ready system maintains continuous evidence through controlled workflows, approvals, and audit trails. Records are captured as activities occur rather than assembled reactively. This reduces audit preparation effort and improves confidence during assessments.
-
How does Pyraman support ISO and management system requirements?
Pyraman integrates document control, audit management, corrective actions, training, and compliance tracking into a single system. This supports structured workflows, preserves traceability, and helps organisations maintain audit readiness as part of normal operations





