From Paper to Digital: How Manufacturing Companies Are Transforming Quality Management

Digital transformation is revolutionizing the manufacturing industry, providing new levels of efficiency, control and competitiveness. While production technologies have significantly advanced, quality management in various organizations has nonetheless relied on paper-based processes, manual tracking and disconnected systems. This gap is now driving manufacturers to reconsider quality management across the whole operation.
Traditional approaches built on physical records, spreadsheets and manual coordination are no longer sustainable in a fast-paced and compliance-driven environment. The move toward paperless management is enabling organizations to eliminate inefficiencies, reduce errors and create a more structured and accessible approach to handling quality data. At the same time, the adoption of business management software is allowing manufacturers to integrate quality processes with broader operational systems, improving visibility and control.
As part of the broader digital transformation in manufacturing industry, organizations are shifting to digital quality management Software to bring uniformity, traceability and real-time visibility into their quality operations. This transition is not simply a matter of exchanging paper for digital resources, it is creating a connected, agile quality management system that drives sustainable operating effectiveness.
Where Paper-Based Quality Systems Fail in Manufacturing Environments
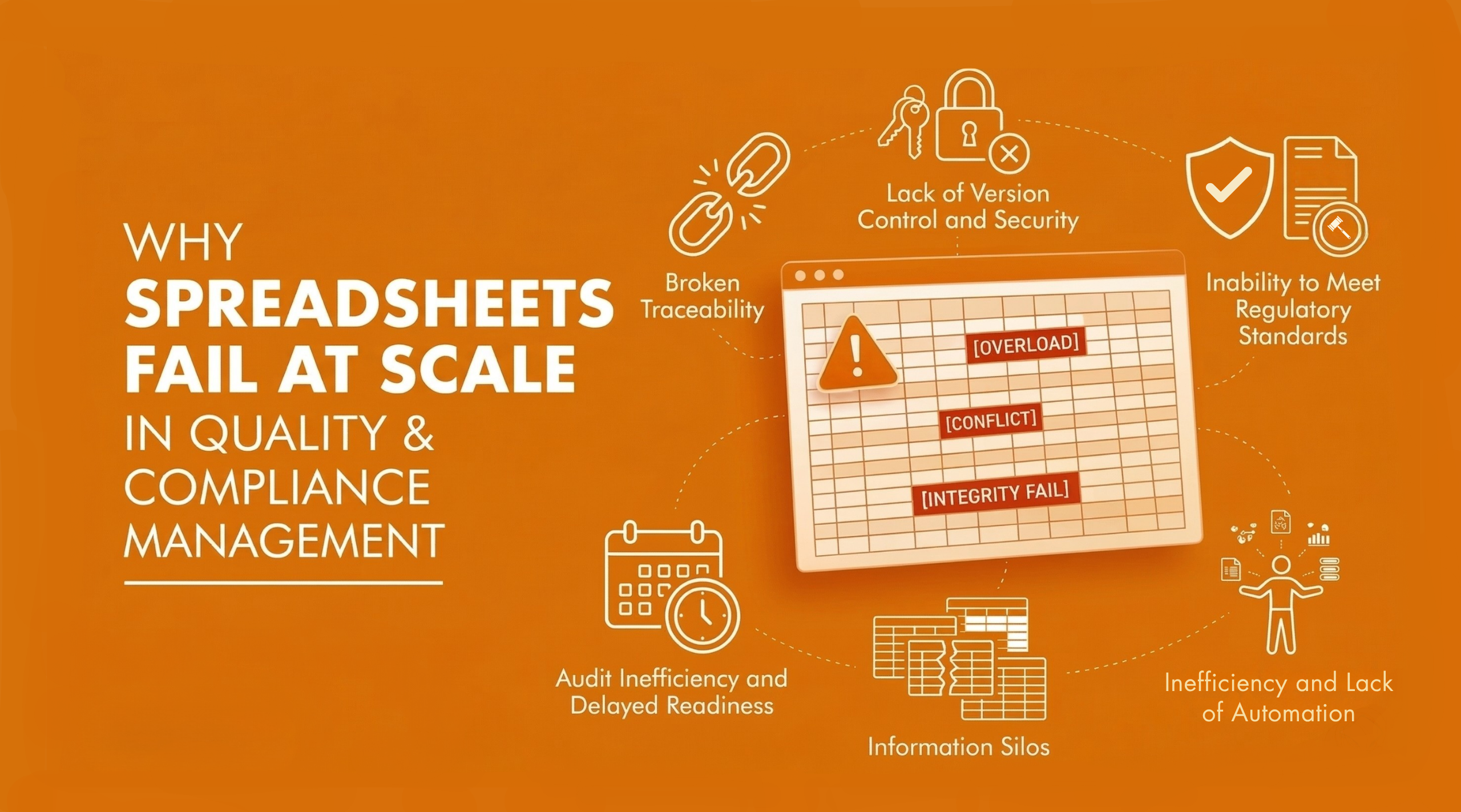
Paper-based quality systems are increasingly unable to support the needs of modern manufacturing operations. In the context of quality management in manufacturing, reliance on manual data entry, physical documentation and disconnected tools makes them difficult to manage, especially as operations scale.
One of the key limitations is the lack of timely access to information. When data is stored across files, spreadsheets and isolated systems, teams end up wasting time searching for documents, which delays the identification and resolution of quality issues. This often leads to slower response times and increased risk of non-compliance. Version control also becomes a challenge, as manually updated documents make it difficult to ensure that teams are working with the most current information. This can result in inconsistencies in processes and outputs.
Tracking approvals, audit trails and corrective actions requires significant manual effort, making the system inefficient and difficult to maintain. These systems also limit collaboration across departments, as information is not centrally available. Teams rely heavily on follow-ups, emails and manual coordination, which slows down communication and decision-making. As manufacturing operations grow in complexity, these limitations highlight the need for a more structured and digitally driven approach to quality management.
How Manufacturing Companies Are Transitioning to Digital QMS
Manufacturing firms are choosing to digitize their QMS one step at a time. Their focus is to replace manual systems with digital systems to create a better impact with less disruption.
-
Assessing Existing Quality Processes
The process begins with the evaluation of the document management systems, audit systems, tracking of non-conformance, approval workflows and systems to identify and find the manual-intensive processes and identify the gaps and inefficiencies.
-
Digitizing Documents and Records
Digitization of records and documentation is a significant and first step. Manufacturers are moving physical documents, records, quality manuals, SOPs, compliance documentation, spreadsheets and records into a single centralized digital system that provides version control and access control.
-
Defining Structured Workflows
Once an organization has a complete digitized system of records, the firm can proceed to the quality processes that include audits and other quality systems, such as non-conformance, CAPA and approval processes. Each of the processes should be mapped and include clear steps, timelines and role assignments.
-
Implementing Workflow Automation
After processes are standardized, companies are now able to automate quality management processes that include the assignment of roles, notification systems and approval systems. This ensures smooth execution of quality processes.
-
Integrating Quality Across Functions
Integration of EQMS to other operational functions such as production, supplier management and training provides quality staff with seamless access to information. Separation of data and information is eliminated between quality and the other operational functions.
-
Establishing Real-Time Monitoring and KPIs
Manufacturers implement dashboards and reporting tools to track quality metrics such as audit status, defect trends and CAPA progress. This enables continuous monitoring and quicker decision-making.
-
Driving Adoption and Continuous Improvement
The final stage is training teams, assuring adoption of the systems and improving the systems continuously, utilizing data and performance driven insights.
With this step-by-step transition, manufacturers move from siloed and reactive systems of quality to integrated and proactive systems of quality in a Digital Quality Management Software.
Top Quality Processes Manufacturers Are Transforming with Digital QMS
Structuring core quality processes digitally creates an integrated and streamlined digital environment for quality management. Some of the core processes that are being digitally transformed are:
-
Document Management System
Quality documents like SOPs, manuals and policies are controlled in a centralised system that has access restrictions, version tracking and approval workflows. This is to ensure that only the most up-to-date approved documents are accessible for use, minimizing inconsistencies and errors. And it makes updating, approving and preparing documents for audits much easier by maintaining clear revision history and access control.
-
Record Management System
Digital filing systems for inspection reports, production records and compliance documentation make them easily accessible. This removes the reliance on physical files and disparate storage solutions. Manufacturers can access historical data within seconds when they have structured record management saving them time and ensuring compliance preparedness during audits, investigations or performance reviews.
-
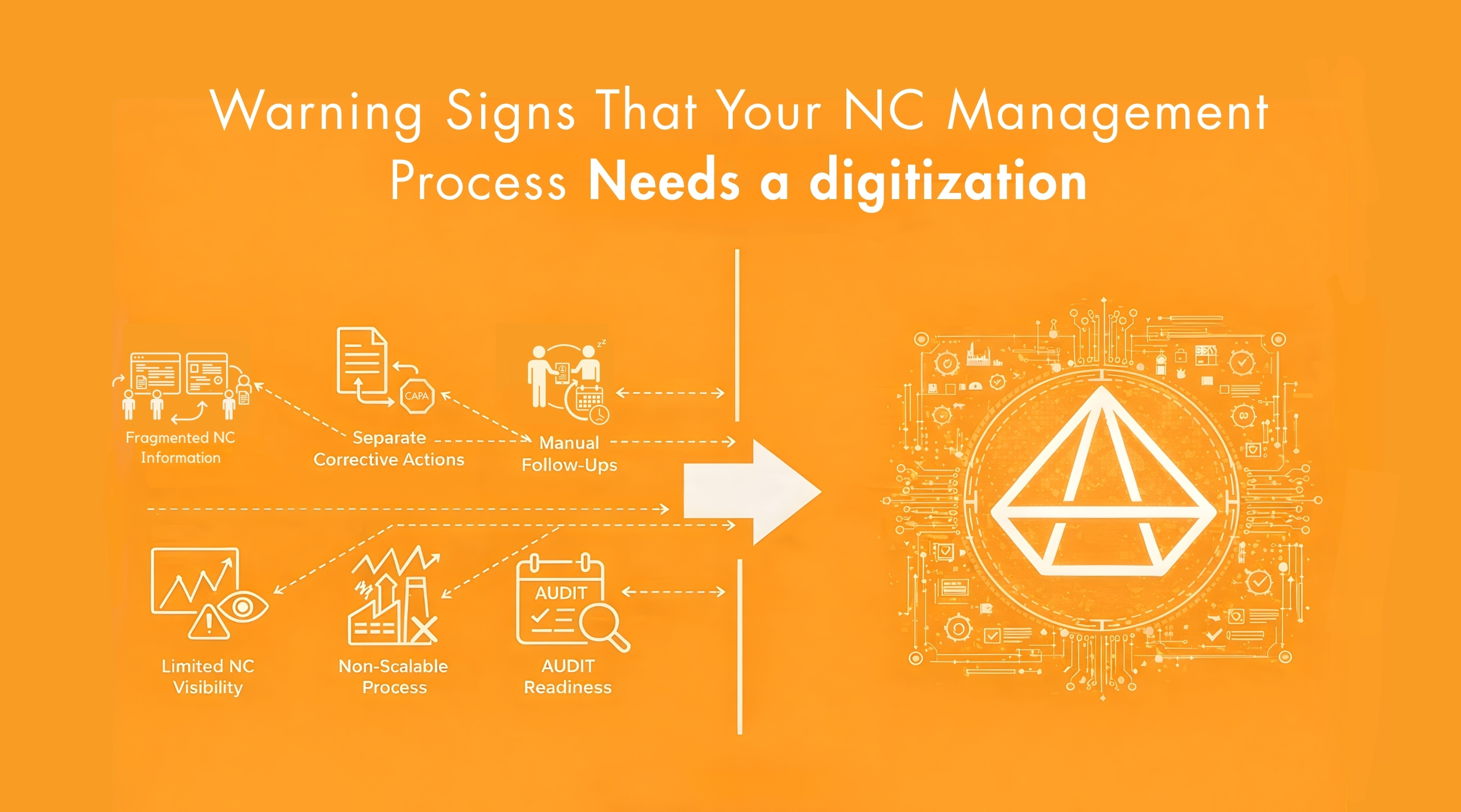
Non-Conformance (NC) Management System
Non-conformities in quality are tracked and processed using digitized workflows, guaranteeing that each issue is documented, investigated and resolved. It enables faster identification of problems, standardized root cause analysis and timely corrective actions as well as corrective actions taken in a timely manner. This minimizes the chance of repetitive problems and ensures product quality remains consistent.
-
Audit Management System
Digital QMS simplifies the end-to-end audit lifecycle, planning, scheduling, execution and reporting. It Increased use of standardized checklists, automated tracking of findings and real-time visibility into audit status can help organizations stay audit-ready at all times. It also helps ensure that observations are adequately resolved and closed in prescribed timeframes.
-
Complaint Management System
Customer complaints flow through defined workflows where they are systematically recorded, tracked and resolved. This ensures quick response, proper investigation and consistency in handling of issues. With full traceability of complaints, manufacturers can easily monitor any recurring issues and implement corrective actions to ensure customer satisfaction.
-
Training Management System
A digital system tracks employee training, skill requirements and certifications. It guarantees sufficient and qualified training to the employees who are engaged in Quality critical processes. It allows organizations to see clear records for audits and regulatory requirements.
-
Objective Management System
Quality objectives are set, monitored and aligned with the business goals. Digital tracking enables organizations to measure performance relative to targets, assign ownership of results and drive accountability. This helps with continuous improvement, as it gives high visibility to progress and results.
-
MoM (Minutes of Meeting) Management System
Meeting discussions, decisions and action points are systematically recorded and tracked within the system, ensuring they are not overlooked or forgotten. This strengthens accountability and ensures that all quality-related actions are followed through within defined timelines.
By digitizing these connected processes with Digital QMS, manufacturers evolve from broken and reactive quality management to a more systematic, transparent and scalable system that enables sustainability in operational excellence.
Benefits of Transitioning to Digital Quality Management
As manufacturing environments become increasingly sophisticated and regulated, the move to a Digital Quality Management System (QMS) creates tangible business outcomes that go beyond traditional process improvement. By enabling paperless management, it can help manufacturers to transition from reactive quality management to a more organization-led and proactive approach for ensuring product quality.
-
Reduced Cost of Quality
Digital QMS helps identify defects early and ensures that issues are addressed through structured workflows before they escalate. By improving root cause analysis and preventing recurrence through CAPA processes, manufacturers can significantly reduce costs associated with rework, scrap, product recalls and warranty claims. Over time, this leads to more predictable and controlled production outcomes.
-
Improved Compliance and Audit Readiness
With centralized documentation, automated approvals and complete audit trails, Digital QMS simplifies compliance management. All records are maintained in a structured and easily accessible format, enabling faster audit preparation and reducing the risk of missing or outdated information. This ensures organizations remain consistently prepared for regulatory and certification audits.
-
Faster Issue Identification and Resolution
Digital systems enable real-time tracking of non-conformities, complaints and quality events, allowing teams to identify and respond to issues more quickly. Automated task assignments and workflow tracking ensure accountability, reduce delays in corrective actions and minimize the impact of quality issues on production.
-
Enhanced Visibility and Data-Driven Decision Making
A key advantage is real-time dashboards and performance insights, enabling management to gain a clear view of quality metrics, trends and risks at any time with EQMS software. This enables organizations to step away from reactive decision-making based on stale reports and instead leverage real-time data to take timely action that enhances overall operational outcomes.
-
Increased Operational Efficiency and Process Standardization
Digital QMS removes the irrelevant tasks, such as manual documentation, repetitive coordination and reliance on spreadsheets for completing quality processes in a department. Standardization of workflows ensures the same approach is followed every time leading to lesser process deviations and higher efficiency for quality and production teams.
-
Improved Traceability, Accountability and Collaboration
Every action, update & approval is tracked in the system maintaining full traceability of quality events. This makes cross-team accountability stronger and prevents issues from being delayed without clear ownership. Also centralized access to information enhances interdepartmental coordination between production, quality and management facilitating better problem-solving.
Adopting Digital Quality Management allows manufacturers not only to enhance their compliance and efficiency but also establish a more resilient, transparent and scalable quality ecosystem that drives sustainable growth.
How Pyraman Helps Manufacturers Transition to Digital QMS
Ready to move beyond fragmented quality systems and implement a structured Digital QMS that delivers real operational control? Pyraman helps manufacturing companies streamline quality processes, improve traceability and scale efficiently without increasing complexity. As part of a modern business management software approach, it ensures that quality management is fully integrated with your operational workflows.
With real-time visibility into quality metrics and standardized workflows, your teams can reduce errors, respond faster to issues and maintain consistent compliance across operations. This means fewer disruptions, better product quality and stronger customer confidence.
Book a personalized demo with Pyraman today to see how digital quality transformation can be implemented within your operations and start driving measurable improvements from day one.
FAQs
-
What is Digital Quality Management Software in manufacturing?
Digital QMS is a software system that manages quality processes like document control, audits, CAPA and compliance. It replaces paper-based work with automated, digital workflows and real-time data.
-
Is Digital QMS a fit for small and mid-sized manufacturers?
Certainly. Digital QMS can be used by small and mid-sized manufacturers. Modern solutions can be adapted and scaled to fit any size business and any individual requirements. They allow smaller businesses to streamline their processes, stay compliant and compete without adding extra operational complexity.
-
How does Digital QMS improve production quality?
Digital QMS software helps improve production quality in part by providing real-time information and data related to production processes and quality metrics. Digital QMS helps identify defects in production earlier, helps to ensure procedures are followed creates documented processes that help improve the speeds at which corrective actions are determined and implemented and allow manufacturers to consistently control the production processes.
-
Will my team need extensive training to use an EQMS?
No. Most modern EQMS platforms are designed for usability at the shop-floor level. With intuitive dashboards and workflow-based systems, training is usually minimal. Adoption becomes easier when the system mirrors your existing processes rather than forcing new ones.
-
Can a digital EQMS integrate with our existing ERP or production systems?
Yes. A good EQMS should integrate seamlessly with your ERP, MES or other operational systems. This ensures data flows automatically between quality, production and compliance functions eliminating silos and manual duplication.
-
Is EQMS Software only useful for large manufacturing companies or can SMEs benefit too?
SMEs often benefit even more because they typically rely heavily on manual systems. A digital EQMS software helps them scale operations, meet compliance requirements and compete with larger players without significantly increasing overhead.
-
Can Digital QMS integrate with existing manufacturing systems?
Yes. Most Digital QMS solutions can integrate with ERP, MES and other systems, enabling seamless data flow and better coordination across production and quality functions.
-
How does Digital Quality Management Software improve audit readiness?
Digital QMS makes audit preparation seamless by storing all documents and records in a centralized system. It maintains complete audit trails, ensuring transparency and accountability. With quick access to real-time data, manufacturers can respond faster to audit requirements. This reduces last-minute stress and ensures smoother, more efficient audits.