Quality Objectives Explained: How a QMS Software Turns Goals into Measurable Results

Quality objectives are a formal requirement within any structured Quality Management System. They are defined to translate quality policy into measurable commitments that guide organisational performance. The alignment between quality policy and objectives ensures that strategic intent is directly reflected in operational performance indicators. When properly governed, they establish direction, accountability and measurable improvement.
However, in many organisations the quality objectives remain documented statements rather than continuously controlled performance indicators. They are reviewed periodically during management meetings or audit preparation, while day-to-day tracking remains fragmented across spreadsheets, reports and departmental records.
Objectives may be clearly written and aligned with policy, also the performance visibility depends on manual consolidation, individual follow-ups or retrospective reporting. Over time, this creates inconsistency in monitoring, limited trend visibility and reactive rather than structured performance control.
Understanding quality objectives therefore requires more than defining them correctly. A clear quality objectives definition establishes how measurable commitments are derived from policy direction and translated into structured performance governance. It requires examining how they are measured, how KPIs support them and how governance determines whether they function as active control mechanisms or remain static compliance documentation.
SMART Quality Objectives: Structuring Measurable Targets
Quality objectives must be structured in a way that enables consistent monitoring and governance without defined parameters, objectives remain interpretive and difficult to control across departments and review cycles.
The SMART principle establishes that objectives should be:
- Specific in scope and clearly defined.
- Measurable through quantifiable indicators.
- Achievable within operational and resource constraints.
- Relevant to organisational priorities and quality policy.
- Time-bound within a defined review period.
These principles collectively form the foundation of smart quality objectives, ensuring that targets remain structured, measurable and governable across review cycles.
When measurable criteria are absent, performance evaluation tends to become subjective and corrective actions are taken without a clearly defined reference point. A structured objective management system brings clarity by establishing a measurable baseline, defining a target level of performance and setting a specific timeframe for review. Together, these elements create the foundation needed for consistent monitoring, meaningful comparison and informed decision making. Such as:
Objective: Reduce internal defect rate
- Baseline: 5%
- Target: Below 1.5%
- Timeframe: Within two quarters
This structure transforms intent into a measurable commitment. Such structured objectives are established; organisations must determine how these targets will be monitored, reviewed and governed regularly.
Understanding KPIs in Quality Objective Governance
Quality Key Performance Indicators (KPIs) are measurable values used to assess how effectively an organisation is achieving its defined quality objectives. While quality objectives define the target outcome the KPIs determine how progress toward that outcome is monitored and evaluated. Without clearly defined KPIs quality objectives cannot be measured consistently, measurable performance requires defined indicators, structured tracking and periodic review.
Quality KPIs are typically categorised into the following areas:
1. Product Quality KPIs:
Product Quality KPIs measure product conformity and adherence to defined specifications. These indicators typically include defect rate, rejection rate and first-pass yield, all of which provide measurable visibility into how consistently products meet quality requirements.
2. Process Quality KPIs:
Process Quality KPIs track process performance, stability and operational efficiency. Metrics such as cycle time, process capability and rework percentage help assess whether processes are controlled, repeatable and performing within acceptable limits.
3. Compliance and Regulatory KPIs:
Compliance and Regulatory KPIs monitor adherence to legal, regulatory and standard requirements. Indicators such as audit findings, CAPA closure rate and inspection outcomes provide measurable oversight into regulatory conformity and corrective action effectiveness.
4. Risk and Safety KPIs
Risk and Safety KPIs evaluate how effectively operational and product risks are managed. These may include incident frequency, risk mitigation implementation rate and safety deviation tracking, offering visibility into preventive control and risk reduction performance.
5. Supplier Quality KPIs:
Supplier Quality KPIs assess supplier reliability and raw material conformity. Metrics such as supplier defect PPM and on-time delivery performance help determine whether supplier performance supports overall quality objectives.
6. Customer Satisfaction KPIs:
Customer Satisfaction KPIs measure customer experience and service quality. Common indicators include complaint rate, complaint recurrence and customer satisfaction score trends, all of which reflect how well organisational outputs meet customer expectations.
7. Continuous Improvement KPIs:
Continuous Improvement KPIs track the implementation and effectiveness of improvement initiatives. Examples include corrective action effectiveness rate and improvement cycle time, which indicate whether structured improvements are delivering measurable results.
Each category supports different quality objectives, but all serve the same purpose converting defined goals into measurable performance indicators. Collectively, these indicators also support the broader objectives of quality control by ensuring systematic oversight of product conformity, process stability and regulatory adherence. Once KPIs are defined, the critical question shifts from what to measure to how those measurements are tracked, consolidated and governed across the organisation.
How Quality Objectives Are Measured in Practice
To understand how quality objectives are measured in practice, it is useful to examine one complete example from definition to performance review. Consider the following structured objective:
Quality Objective:
Reduce internal defect rate from 3.5% to below 1.5% within two quarters.
This objective is measurable because it defines:
- A baseline (3.5%).
- A target threshold (below 1.5%).
- A defined timeframe (two quarters).
However, the objective alone does not ensure monitoring. It must be supported by a clearly defined KPI.
- Defined KPI: Monthly Internal Defect Percentage.
- Calculation Method: (Number of defective units ÷ Total units produced) × 100.
- Data Source: Daily production inspection logs.
- Monitoring Frequency: Consolidated monthly.
- Review Level: Presented in monthly quality review and quarterly management review.
In practice, measurement would function as follows:
- Production inspection teams record daily defect data.
- Data is consolidated at the end of each month.
- The monthly defect percentage is calculated using the defined formula.
- The result is compared against both the baseline (3.5%) and target (1.5%).
- If deviation persists, corrective actions are initiated.
- Trend analysis is reviewed across months to evaluate improvement trajectory.
The objective establishes the performance commitment, while the KPI defines how it is measured, supported by a consistent calculation method and a defined review cycle that ensures ongoing governance.
When such structures exist for a limited number of objectives, tracking may remain manageable. However, as additional objectives are introduced across production performance, compliance metrics, supplier reliability and customer satisfaction, the number of KPIs increases. Each requires data capture, consolidation, comparison and review. At this point, measurement complexity begins to scale.
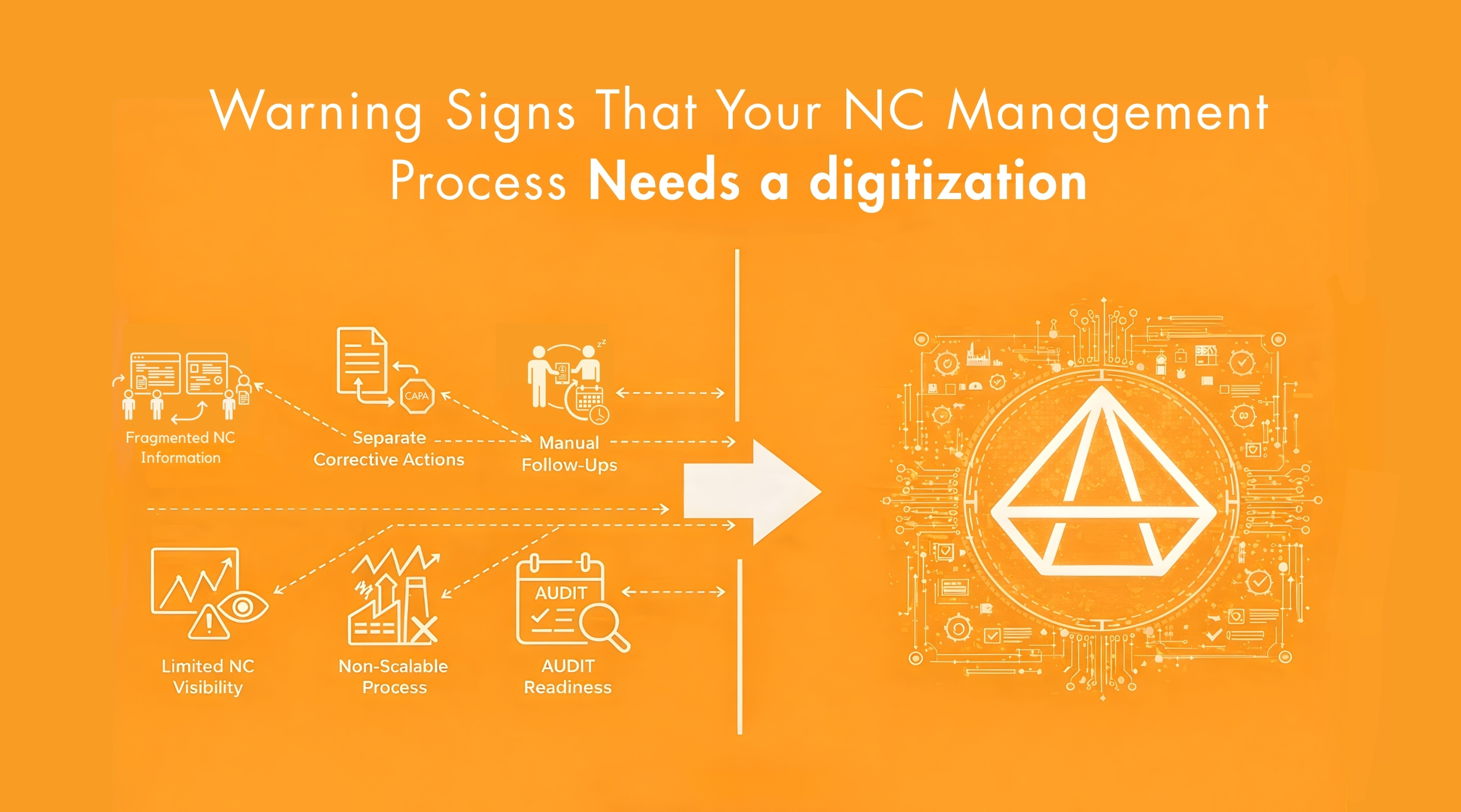
Limitations in Manual Quality Objective Tracking
When quality objectives are limited in number, manual tracking may remain manageable. However, as objective volume increases across departments and operational functions, the tracking framework must scale accordingly. If measurement mechanisms remain manual, structural strain gradually develops. The limitation does not arise from objective definition. It arises from sustaining measurable governance across expanding KPIs. The following conditions typically indicate structural constraint.
-
KPI Records Are Maintained in Separate Files:
KPI data may exist across spreadsheets, production logs, audit trackers and departmental reports. Consolidation requires manual compilation prior to review cycles. Over time, traceability weakens and reporting effort increases.
-
Monitoring Occurs Primarily Before Review Meetings
Performance data is often consolidated shortly before monthly or quarterly management review. Between review periods, KPI visibility remains limited, reducing continuous oversight.
-
Calculation Methods Are Not Centrally Controlled
If KPI formulas, baseline definitions or measurement criteria vary across departments or reporting periods, comparison becomes inconsistent this reduces confidence in trend accuracy.
-
Accountability Depends on Follow-Up
KPI ownership may be understood operationally but not governed structurally. Tracking depends on reminders and coordination rather than workflow enforcement.
-
Trend Analysis Requires Retrospective Compilation
Historical comparison often requires manual extraction of past reports. Recurring deviations may remain undetected until formal review.
-
KPI Volume Expands Without Integrated Oversight
As objectives increase across production quality, compliance performance, supplier management and customer indicators, the number of associated KPIs increases proportionally. Manual coordination effort scales at the same rate, while governance capability does not.
Under these conditions, quality objectives remain documented commitments, but measurable control becomes dependent on reporting activity rather than system structure.
How a Structured QMS Software Turns Goals into Measurable Results
A structured QMS software provides the system foundation required to govern quality objectives in a reliable and scalable manner. Instead of relying on manual consolidation and informal coordination, objective tracking is integrated within controlled workflows. This ensures that measurable oversight is maintained consistently, regardless of objective volume, departmental scope or operational complexity. A structured QMS environment typically enables the following controls.
-
Controlled Objective Definition with Linked KPIs
Quality objectives are defined within a centralised framework where baseline values, target thresholds and review periods are formally recorded. Each objective is directly linked to its associated KPI, ensuring that performance commitments and measurement logic remain integrated.
-
Standardised KPI Configuration with Defined Calculation Logic
KPI formulas, measurement parameters and data sources are configured within the system. This prevents variation in calculation methods across departments or reporting cycles and ensures that performance comparison remains consistent over time.
-
Integrated Ownership and Monitoring Workflows
Responsibility for KPI monitoring is assigned within structured workflows. Status updates, deviations and performance thresholds are tracked within the system, reducing dependence on follow-up reminders or manual reporting coordination.
-
Performance Visibility Through Integrated Dashboards:
Performance data is consolidated automatically within defined dashboards. Target comparison, baseline alignment and trend progression remain visible in real time, enabling ongoing oversight rather than periodic compilation.
-
Integrated Linkage Across Quality Processes
Quality objectives are connected to related quality activities such as non-conformance management, corrective actions and audit findings. When deviations occur, traceability is maintained between the KPI, the objective and the associated corrective action, preserving governance continuity.
By integrating objective definition, KPI configuration and performance tracking within a structured system, QMS software transforms quality objectives from static documentation into continuously governed performance mechanisms. Measurement becomes integrated rather than coordinated. Oversight becomes system-driven rather than report-dependent. As objective volume increases, governance strength is sustained rather than diluted.
How Pyraman Supports Measurable Quality Objectives
Quality objective governance defines how performance commitments should be measured and reviewed, but its effectiveness depends on how consistently those controls are applied in practice. Pyraman supports organisations by enabling objective management through a structured system that integrates definition, KPI configuration and performance tracking within a controlled environment.
Pyraman replaces fragmented KPI spreadsheets and periodic reporting practices with a unified objective management system. Quality objectives are defined, linked to associated KPIs and monitored within a single system, ensuring that measurable oversight is applied consistently across departments, locations and operational functions.
By enforcing defined ownership, standardised calculation logic and structured review workflows, Pyraman ensures that performance measurement remains controlled rather than interpretive. KPI tracking is system-driven rather than dependent on follow-up reminders or manual consolidation, reducing variability as objective volumes increase.
Pyraman also strengthens governance by maintaining continuous visibility into baseline alignment, target progression and trend history. Performance records remain traceable across review cycles, supporting audit readiness and management oversight without reliance on retrospective compilation.
Through integrated objective management, Pyraman enables quality objectives to function as continuously governed performance mechanisms rather than static documentation reviewed only during formal meetings.
FAQs
-
How does Pyraman help track quality objectives across multiple departments?
Pyraman centralises quality objectives within a unified digital framework that allows departments to monitor their assigned KPIs in one system. Each objective is linked to responsible owners and defined review cycles. This eliminates fragmented tracking across spreadsheets. Cross-department visibility ensures consistent governance and aligned performance monitoring.
-
Can Pyraman link quality objectives directly to KPIs and corrective actions?
Yes, Pyraman connects quality objectives directly with their associated KPIs and related corrective actions. When deviations occur, the system maintains traceability between performance data and CAPA workflows. This ensures that improvement actions are structured and measurable. Governance remains integrated rather than disconnected.
-
How does Pyraman reduce manual KPI consolidation effort?
Pyraman automates KPI data capture and consolidation within a centralised dashboard environment. Instead of manually compiling reports before review meetings, performance metrics are updated in real time. This reduces administrative effort and reporting errors. Management can access structured performance insights without retrospective compilation.
-
Can Pyraman provide real-time dashboards for quality objective monitoring?
Yes, Pyraman offers configurable dashboards that display baseline alignment, target progress and trend history in real time. Performance visibility remains continuous rather than periodic. Decision-makers can identify deviations early and initiate corrective action promptly. This strengthens proactive performance governance.
-
How does Pyraman improve audit readiness related to quality objectives?
Pyraman maintains documented traceability between objectives, KPIs, review records and corrective actions. Historical performance data remains accessible and structured for audit verification. This reduces last-minute preparation and manual evidence collection. Audit readiness becomes system-driven rather than reactive.
-
Is Pyraman suitable for multi-site quality objective tracking?
Yes, Pyraman supports multi-site governance by consolidating objective tracking across locations within a single platform. Standardised KPI definitions ensure consistency in performance measurement. Central oversight remains intact while allowing site-level accountability. This supports scalable quality management across distributed operations.
-
How does Pyraman standardise KPI calculation across departments?
Pyraman allows predefined KPI formulas and measurement parameters to be configured centrally. This prevents variation in calculation methods across departments or reporting periods. Consistent logic ensures reliable trend analysis and comparison. Standardisation strengthens confidence in reported performance data.
-
Can Pyraman scale as the number of quality objectives increases?
Yes, Pyraman is designed to scale with organisational growth and expanding objective volumes. As new KPIs and departments are added, governance remains structured within the same controlled framework. The system prevents tracking complexity from overwhelming oversight capability. Performance control strengthens rather than dilutes over time.
-
How does Pyraman ensure accountability in quality objective monitoring?
Pyraman assigns defined ownership for each objective and KPI within structured workflows. Status updates, review cycles and deviations are recorded within the system. Automated notifications reduce dependency on follow-up reminders. Accountability becomes embedded within the process rather than informal coordination.
-
What makes Pyraman different from spreadsheet-based quality tracking?
Unlike spreadsheets, Pyraman integrates objectives, KPIs, corrective actions and review workflows within one controlled environment. It provides real-time visibility, traceability and structured governance. Manual consolidation and version control issues are eliminated. Performance monitoring becomes systematic rather than report-driven.